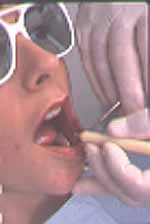
 Future so bright: Patients as
well as dental team members must wear special glasses to protect their
eyes from the laser light.
Future so bright: Patients as
well as dental team members must wear special glasses to protect their
eyes from the laser light. |
Dental Lasers Approved by FDA
 Future so bright: Patients as
well as dental team members must wear special glasses to protect their
eyes from the laser light.
Future so bright: Patients as
well as dental team members must wear special glasses to protect their
eyes from the laser light. |
FDA approves first dental laser system for treating decay
Laser company expects to sell 10,000 within the next decade
"FDA approves 'painless' laser drill for teeth," blares Thursday's headline in The Wall Street Journal (WSJ).
The WSJ, along with the rest of the general media, picked up on a news release issued May 7 by the Food and Drug Administration announcing that the federal agency had cleared for marketing the first laser system for treating tooth decay for use directly on teeth. Cleared for marketing was the Centauri Erbium Yttrium Aluminum Garnet, or Er:YAG, laser system made by Premier Laser Systems Inc. of Irvine, Calif.
|
"Use of lasers in dentistry is medicine for the 21st Century," Bruce Burlington, M.D., director of FDA's Center for Devices and Radiological Health, said in the agency's news release. "They will give dentists and patients a significant new option for treating decay and doing other related procedures."
Dental lasers previously cleared by the FDA are for use only on soft tissue such as the gums.
The FDA broke the news to dentistry and the general public at the same time, as is its policy. The federal agency is prohibited by law from releasing or discussing the status of any company's application, said Sharon Snider, an FDA spokesperson.
"It's considered trade secret information," she added. "Once a product has been approved, then all that information becomes releasable.
"And the FDA had just approved Premier's laser for hard tissue applications as of Wednesday (May 7)," Ms. Snider noted.
Armed with clearance to market its laser, Premier predicted it may sell 10,000 to 15,000 Er:YAG laser systems within the next decade.
In its press release issued Thursday, Premier noted that the laser system could be in "dentists' hands within the next 90 days." Dentists who do purchase the laser are required to participate in a training program provided by Premier.
Though the laser system "virtually eliminates" the need for anesthesia in appropriate hard tissue applications, according to Premier, don't look for dental fees to drop just yet.
Noted the WSJ, "The lasers could bring higher prices for fillings and other dental restorations." Why? For the simple reason the laser comes with a $39,000 price tag.
The ADA, in a prepared statement released Thursday, described the FDA action as promising. However, since Premier and the FDA have not provided the ADA with the laboratory and clinical studies on which this decision was made, the Association cannot attest to the safety and effectiveness of this laser application at this time.
Some of the concerns that the Association has raised in the past regarding the use of lasers on hard tissue include:
The ADA added that a comprehensive statement will be published after the Association has reviewed studies demonstrating the safety and effectiveness of lasers for hard tissues.
In its news release, the FDA cited studies conducted by Premier that showed the Er:YAG laser "is as safe and effective as a high speed drill for removing dental decay and preparing a cavity for a filling."
Fewer patients, the FDA added, needed anesthetic for pain according to Premier's study.
The Er:YAG also may be used to roughen enamel to improve bonding of a restoration, the FDA said.
Premier conducted two clinical studies. In the first, according to the FDA, more than 500 teeth with decay were treated with the laser and later evaluated for damage to the tooth's nerve and blood supply. No adverse events were reported.
Of the 125 patients with decay enrolled in the second study, half were treated with the laser and the other half were treated with a high speed drill. Results revealed that the laser was "as safe and effective as the drill in removing decay and preparing the tooth for a filling," the FDA said.
Dr. Richard T. Hansen, a faculty member at the University of California at Los Angeles dental school who participated in Premier's clinical studies, said the results of his and other patient studies "underscored the fact that a laser for hard tissue procedure writes a new page in dental history," according to the manufacturer's press release.
"We can pinpoint and excise diseased hard tissue with great precision, preserving the tooth's overall integrity," Dr. Hansen continued.
The laser system consists of a box-like laser console with a fiber optic cable connected to a dental handpiece, which resembles a standard high-speed drill. The laser uses water to cool the tooth and clean the surface during removal of decay and cavity treatment.
Dentists and patients will be required to wear goggles during procedures to protect their eyes, the FDA added.